
Stool samples for microbiome analyses .
The OMNIgene®-GUT kits OM-200 and OMR-205 fulfill all requirements for best sample quality and a realistic microbiome profile: The sampling itself is simple and clean, perfect for self-sampling by patients or study participants. The stool sample is immediately homogenized in the tube, and the preservation solution stabilizes nucleic acids very effectively. This genetic snapshot of the microbiota is preserved for a long time even without refrigeration during storage and transport.
Hard Facts .
- Intuitive and user-friendly sample collection
- Minimization of microbial growth
- Minimization of DNA degradation
- Stabilization of DNA during temperature fluctuations (-20°C to 50°C)
- Very good signal-to-noise ratio in subsequent data analyses

Applications .
Perfect sample homogenization and stabilization is necessary to minimize falsification of the gut microbiome. A wide variety of microbes can be reliably detected from the samples obtained using downstream applications.
Macklaim, J., Tayeb, M., Dillane, C., Fragel, C., & Cunningham, L. (2021). The need for stabilization: short-term variation in microbiome profiles. DNA Genotek https://www.dnagenotek.com/ROW/pdf/PD-WP-00070.pdf
Doukhanine, E., Bouevitch, A., Pozza, L., & Merino, C. (2014). OMNIgene®-GUT enables reliable collection of high quality fecal samples for gut microbiome studies. DNA Genotek https://www.dnagenotek.com/ROW/pdf/PD-WP-00040.pdf
Doukhanine, E., Bouevitch, A., Brown, A., Gage LaVecchia, J., Merino, C., & Pozza, L. (2016). OMNIgene®-GUT stabilizes the microbiome profile at ambient temperature for 60 days and during transport. DNA Genotek https://www.dnagenotek.com/ROW/pdf/PD-WP-00042.pdf
Brown, A., Lynch, D., Bouevitch, A., & Doukhanine, E. (2018). OMNIgene®-GUT provides easy self-collection and stabilization of liquid fecal samples for microbiome profiling. DNA Genotek. https://www.dnagenotek.com/ROW/pdf/PD-WP-00056.pdf
Tayeb, M., Macklaim, J., Colemeco, S., Fragel, C., Dillane, C., & Cunningham, L. (2020). Impact of fecal collection methods on sample homogenization and extraction reproducibility in gut microbiome profiling. DNA Genotek https://www.dnagenotek.com/ROW/pdf/PD-WP-00064.pdf
16S rRNA analyses enable the detection, identification and characterization of the microorganisms of the gut microbiome.
Anderson, E. L., Li, W., Klitgord, N., Highlander, S. K., Dayrit, M., Seguritan, V., Yooseph, S., Biggs, W., Venter, J. C., Nelson, K. E., & Jones, M. B. (2016). A robust ambient temperature collection and stabilization strategy: Enabling worldwide functional studies of the human microbiome. Scientific Reports, 6(1), 31731. https://doi.org/10.1038/srep31731
Choo, J. M., Leong, L. E., & Rogers, G. B. (2015). Sample storage conditions significantly influence faecal microbiome profiles. Scientific Reports, 5(1), 16350. https://doi.org/10.1038/srep16350
The human gut microbiome is increasingly becoming the focus of research. There is increasing evidence that the microbiome plays a crucial role in many physiological, pharmacokinetic and pathological processes.
Abrahamson, M., Hooker, E., Ajami, N. J., Petrosino, J. F., & Orwoll, E. S. (2017). Successful collection of stool samples for microbiome analyses from a large community-based population of elderly men. Contemporary Clinical Trials Communications, 7, 158-162. https://doi.org/10.1016/j.conctc.2017.07.002
OMNIgene GUT - OM-200 .
Product overview .
| Attribute | OM-200 | OMR-205 |
|---|---|---|
| Collection site | Feces | Feces |
| Sample homogenization | ✔ | ✔ |
| Microbial nucleic acids | DNA | DNA and RNA |
| Sample collected per kit (median) | 520 ± 101 mg | 545 mg |
| Shipping at ambient temperature | ✔ | ✔ |
| Compatible with a wide variety of nucleic acid extractions and purification methods | ✔ | ✔ |
| Standardized format for high-throughput processing | ✔ | ✔ |
| Number of extractions per kit (250 µL per extraction) | 8 | 10 (200 µl per extraction) |
| DNA yield per extraction ⌀ (median) | 12 ± 1.49 µg (12.56 µg) | 4.40 ± 0.52 µg (2.06 µg) |
| High molecular weight DNA | ✔ | ✔ |
| Microbiome DNA stability at room temperature | 60 days | 30 days |
| Microbiome RNA stability at room temperature | – | 10 days |
| Suitable for NGS downstream application | ✔ | ✔ |
| Suitable for long-term storage and biobanking | ✔ | ✔ |

OM-200 - extended DNA stability .

OMNIgene® GUT is an all-in-one system for easy self-collection of stool samples for gut microbiome profiling for research purposes.

- Improved donor compliance due to intuitive and user-friendly handling
- Easy self-sampling by patients and study participants
- Minimization of distortions caused by microbial growth and DNA degradation.
- Eliminates the need for weighing and aliquoting with a standard volumetric sample.
- The DNA remains stable in the sample tube for 60 days - no cold chain required
- Maintenance of DNA integrity during fluctuations in ambient temperature (e.g. -20 °C to 50 °C).
- Immediate homogenization and stabilization of the sample during collection
- Tube format is suitable for manual or automated processing
- High quality DNA for 16S rRNA profiling, shotgun sequencing, qPCR, arrays
- Barcoding for complete traceability of the samples
Further information .
-
Manufacturer websiteManufacturer website
-
Instruction manual (engl.)Instruction manual (engl.)
OMR-205 .

OMNIgene® GUT OMR-205 is an all-in-one system for easy self-collection of stool samples for DNA and RNA stabilization for gut microbiome profiling. The product is intended for research purposes.

- Improved donor compliance due to intuitive and user-friendly handling
- Minimization of microbiome distortions
- Stability of DNA 30 days and RNA 10 days at room temperature
- Eliminates the need for weighing and aliquoting with a standard volumetric sample.
- Maintains DNA and RNA integrity during ambient temperature fluctuations (e.g. -20 °C to 50 °C).
- Immediate homogenization and stabilization of the sample during collection
- High-quality DNA and RNA for multi-omics downstream analyses
- Tube format is suitable for manual or automated processing
- Barcoding for complete traceability of the samples
Further information .
-
Manufacturer websiteManufacturer website
-
Instruction manual (engl.)Instruction manual (engl.)
All-in-one shipping kit OM-200+ACP102 .

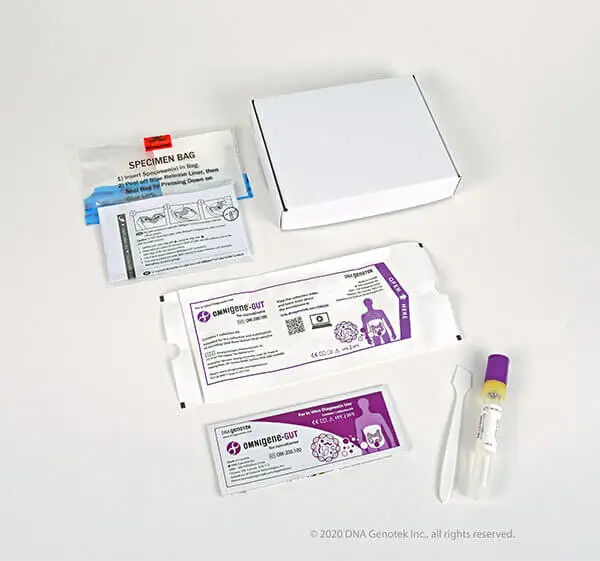
Kit configuration .
- OM-200: OMNIgene-GUT including user manual and spatula
- OM-AC1: Toilet accessories for simplified sampling
- MO-5: Biospecimen bag
- 2-way shipping carton (with peelable adhesive strip for sample return)
- Individual label for traceability
DNA stability .
OMNIgene-GUT-stabilized samples were extracted at baseline (T0) and after 14- and 60-day storage at room temperature (23°C). Paired non-stabilized samples were extracted at baseline (T0, fresh) and after 14 days at room temperature (23°C) or after 14 days at -80°C. Similarity of samples at baseline and after storage was evaluated using Bray-Curtis distances.
From: Doukhanine, E., Bouevitch, A., Brown, A., Gage LaVecchia, J., Merino, C., & Pozza, L. (2016). OMNIgene®-GUT stabilizes the microbiome profile at ambient temperature for 60 days and during transport. DNA Genotek https://www.dnagenotek.com/ROW/pdf/PD-WP-00042.pdf

Bray-Curtis distance difference between unstabilized (14 days at 23°C), OMNIgene-GUT (60 days at 23°C) and -80°C (14 days at -80°C) samples compared to fresh samples. Significant dissimilarity was scored as Mann-Whitney (**P≤0.001).
RNA stability in OMR-205 .

© DNA Genotek, Inc. All rights reserved.
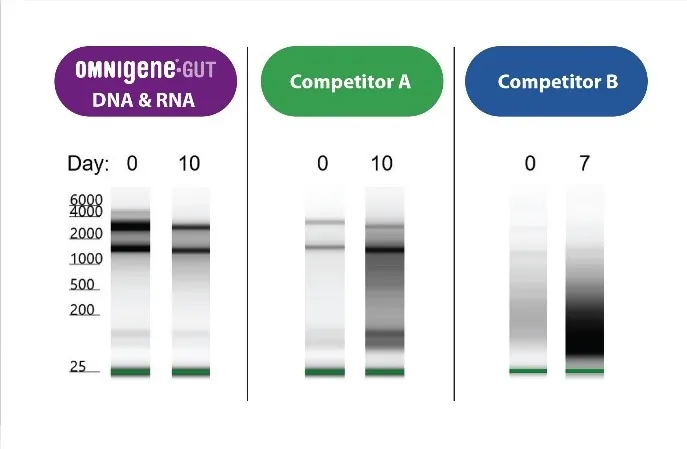
No bias in qPCR .
The compatibility of samples collected with OMNIgene-GUT with the PowerFecal DNA Isolation Kit was tested. A fresh sample and an OMNIgene-GUT sample were collected from the same fecal bulk sample from three different donors. DNA was extracted from an aliquot of each sample and DNA performance was determined by 16S rRNA qPCR.
From: Doukhanine, E., Bouevitch, A., Pozza, L., & Merino, C. (2014). OMNIgene®-GUT enables reliable collection of high quality fecal samples for gut microbiome studies. DNA Genotek. https://www.dnagenotek.com/ROW/pdf/PD-WP-00040.pdf
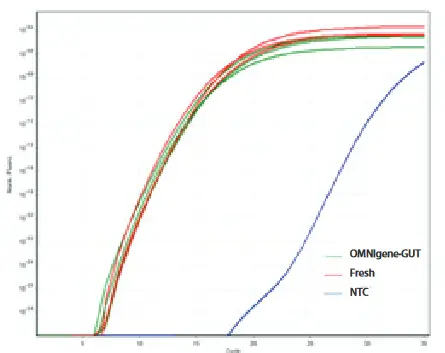
Real-time 16S rRNA PCR of fecal DNA extracted from 3 donors using OMNIgene-GUT or fresh samples (∆∆Cq = 0.54). NTC – no-Template Control.
No shift of the microbiome profile during storage .
Brown, A., Lynch, D., Bouevitch, A., & Doukhanine, E. (2018). OMNIgene®-GUT provides easy self-collection and stabilization of liquid fecal samples for microbiome profiling. DNA Genotek. https://www.dnagenotek.com/ROW/pdf/PD-WP-00056.pdf
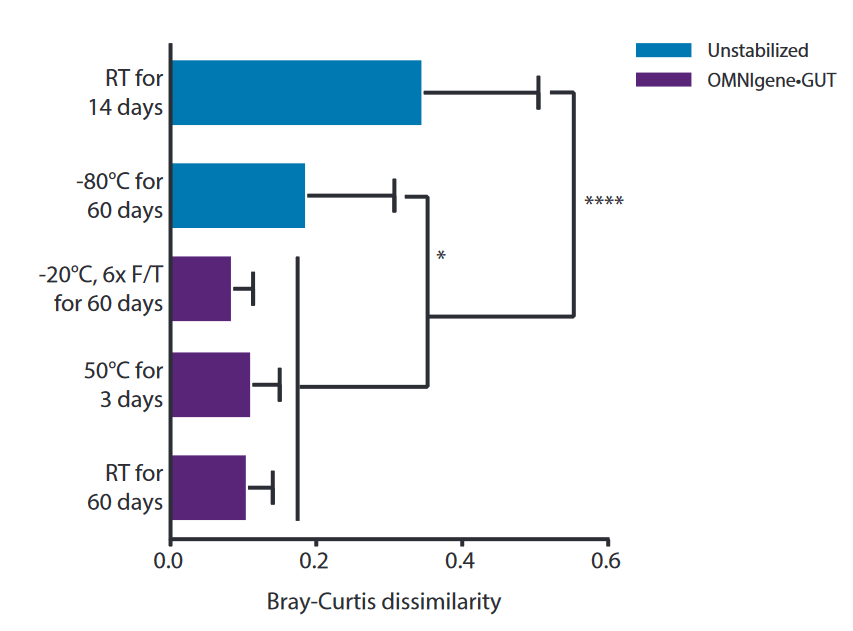
Bray-Curtis dissimilarity of stool samples collected with OMNIgene-GUT from donors with dysbiosis and then exposed to storage and simulated shipping conditions. Dissimilarity was determined by comparing the microbial profile of the sample at baseline with paired samples. A significant difference in microbiome profile was observed in unstabilized (blue) samples stored at RT or -80°C compared to OMNIgene-GUT (purple) stabilized samples (*P≤0.05 and ****P≤0.0005, respectively).
No shift of the microbiome profile during storage .
OMNIgene-GUT samples were stored with OMR-205 for 10 days at room temperature and exposed to simulated transport conditions. Microbiome profile (DNA and RNA) bias is minimal compared to freshly collected samples.

© DNA Genotek, Inc. All rights reserved.
Realistic snapshot of the microbiome .
The samples obtained with OMNIgene kits are of excellent quality. The high integrity of the stabilized DNA allows a very realistic representation of the microbiome profile.
From: DNA Genotek. (n.d.). OMNIgene Devices.
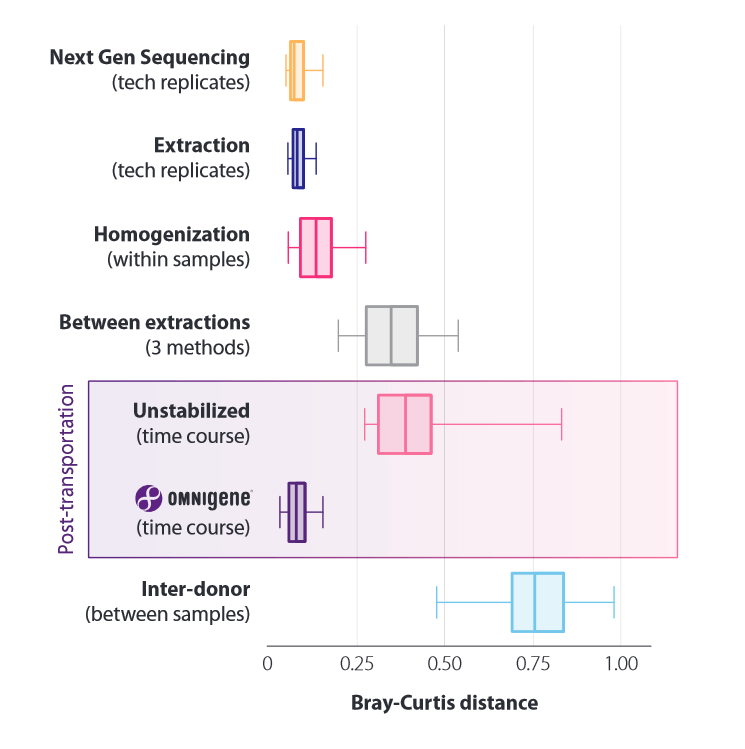
Quantification of the effects of pre-analytical factors: microbiome profile differences between technical replicates during sequencing, extraction, sample homogenization, of 3 different kits in comparison. Analysis was performed with respect to transport (temperature fluctuations between -8°C and +23°C, unstabilized and OMNIgenes) and donor to donor (control).
High stability of the microbiome profile .
During transport from the sample collection to the processing laboratory, the samples are usually exposed to unfavorable conditions. To simulate this, unstabilized samples and samples stabilized with OMNIgene-GUT were stored at 50°C or 37°C for three days or subjected to multiple freeze-thaw cycles.
From: Doukhanine, E., Bouevitch, A., Brown, A., Gage LaVecchia, J., Merino, C., & Pozza, L. (2016). OMNIgene®-GUT stabilizes the microbiome profile at ambient temperature for 60 days and during transport. DNA Genotek. https://www.dnagenotek.com/ROW/pdf/PD-WP-00042.pdf
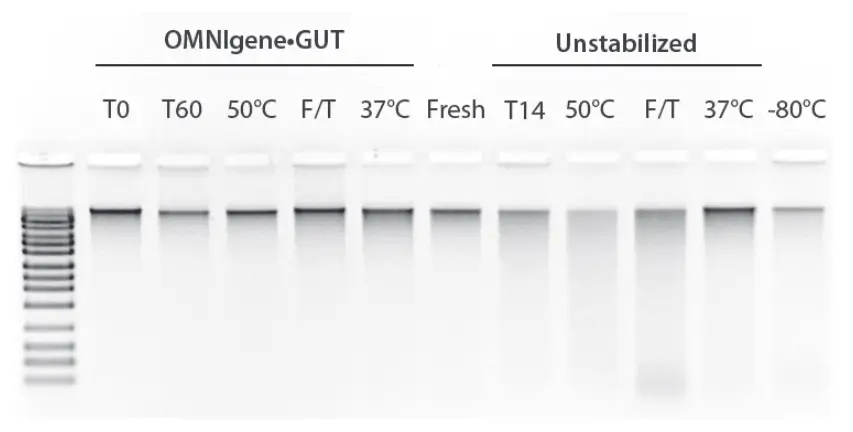
DNA integrity of OMNIgene-GUT samples under simulated transport conditions. OMNIgene-GUT samples from a representative donor were stored at 23°C for 60 days (T60), 50°C for 3 days (50°C), or 37°C for 3 days (37°C); or subjected to 6 freeze-thaw cycles (F/T). Unstabilized samples from the same donor were stored at 23°C (T14) for 14 days, at 50°C (50°C) for 1 day, or at 37°C (37°C) for 3 days, or at -80°C (-80°C) for 14 days.

