Sample collection for the diagnosis of infectious diseases .
The DNA Genotek OMNIgene ORAL or Colli-Pee® kits are easy to handle and therefore also ideally suited for home use. Since the DNA and RNA remain stable in the tube up to +50°C, no cold chain is required.
Hard Facts .
- High sample stability
- Easy handling and home sampling possible
- No cold chain required
- Obtained DNA/RNA is compatible with qPCR and RT-qPCR assays


Applications .
OMNIgene ORAL products provide a painless, non-invasive alternative to blood collection for the detection of circulating Plasmodium falciparum or P. vivax parasites.
Okumu, W., Okeyo, W. A., Munde, E. O., Raballah, E., Anyona, S. B., & Ouma, C. (2015). Evaluation of saliva-derived Plasmodium falciparum DNA using the OMNIgene®-ORAL kit in detection of malaria. DNA Genotek https://www.dnagenotek.com/ROW/pdf/PD-WP-00035.pdf
Visanuvimol, D. (2014). Saliva as a proven, non-invasive sample type for molecular malaria testing and surveillance using OMNIgene®-ORAL at ambient temperatures. DNA Genotek https://www.dnagenotek.com/ROW/pdf/PD-WP-00041.pdf
The gold standard for detecting SARS-CoV-2 infection is the detection of viral RNA in saliva samples or oral, pharyngeal, and nasal swabs by PCR/qPCR. The OMNIgene ORAL Kit OME-505 stabilizes SARS-CoV-2 RNA in collected saliva samples over several weeks at room temperature and allows sample collection at home.
DNA Genotek. (2020). Inactivation of SARS-CoV-2 in samples collected using Oragene®, ORAcollect® and OMNIgene® products from DNA GenotekTM. https://www.dnagenotek.com/ROW/pdf/MK-01430.pdf
Colameco, S., Brown, A., & Crawford Parks, T. (2020). Ambient temperature stabilization of SARS-CoV-2 viral RNA in OMNIgene®-ORAL (OME-505) collected saliva samples. DNA Genotek https://www.dnagenotek.com/ROW/pdf/PD-WP-00069.pdf
Stabilization of viral DNA and RNA after sampling is important to achieve consistent quality results and avoid bias. The OME-505 kit (formerly OM-505) allows long storage time of up to three weeks at room temperature without loss of DNA or RNA quality.
DNA Genotek. (2015). OMNIgene®-ORAL (OM-505) stabilizes RNA from human viruses at room temperature for several weeks. https://www.dnagenotek.com/ROW/pdf/PD-WP-00024.pdf
Doukhanine, E., & Merino, C. (2020). Stability, recovery efficiency and detection limit of viral DNA in OMNIgene®-ORAL (OM-505). DNA Genotek https://www.dnagenotek.com/ROW/pdf/PD-WP-00028.pdf
Molecular detection of human papillomavirus (HPV) DNA in urine is suitable for screening for cervical precancerous lesions. Colli-Pee® enables user-friendly self-collection of urine, eliminating the need for cervical swabs for cytological analysis.
Mehta, A., Laeremans, M., Meers, N., Ríos Cortés, A., Van Avondt, Q., Pasmans, D., Beyers, K., & Vankerckhoven, V. (2021). First-void urine as a game-changer for HPV-based cervical cancer screening. Novosanis. Link
Laeremans, M., Mehta, A., Pasmans, D., Jordaens, S., Faster, D., Beyers, K., & Vankerckhoven, V. (2021). HPV-based cervical cancer screening: gaining insights into sample preference and cost-effectiveness. DNA Genotek. Link
Pasmans, D., Mehta, A., Bruyninckx, S., Hendrickx, J., Beyers, K., & Vankerckhoven, V. (2021). Molecular-based triage on hrHPV-positive women to detect cervical cancer. DNA Genotek. Link
First urine is suitable for detecting pathogen DNA of sexually transmitted infectious diseases (STIs), including human papillomavirus (HPV) and Chlamydia trachomatis (CT).
Mehta, A., Pasmans, D., Vankerckhoven, V., & Beyers, K. (2021). Urine collected through Colli-Pee offers potential for self-sampling at home for detection of sexually transmitted infections. Novosanis. Link
Details .
Various kits for sampling .
| OME-505 | ORE-100 | OM-501 | Colli-Pee® | |
|---|---|---|---|---|
| Nucleic acid | DNA and RNA | RNA | DNA | DNA |
| Collection site | Saliva | Oral cavity | Saliva | Urine |
| Stability | 3 weeks | 60 days | 1 year | < 14 days at RT |
| Swab-based | ✔ | |||
| CE-IVD certified | ✔ | ✔ | ✔ |



OME-505 .
OMNIgene ORAL OME-505 is perfect for the collection and stabilization of microbial nucleic acids. The kit provides excellent sample integrity and stabilization. This is essential to exclude bias when analyzing the microbiome using microarrays or next generation sequencing.

Advantages .
- CE/IVD certified
- Easy self-collection of high-quality DNA and RNA from oral samples
- Stabilizes samples from collection to analysis of DNA and RNA - no refrigerated transport necessary
- Snapshot of the microbial profile at the time of sampling.
- Allows simultaneous detection of DNA and RNA of bacteria and viruses from a single sample
- Reduces transport and storage costs and process complexity due to 3 weeks of room temperature stability of DNA and RNA
- Allows identification of live or metabolically active bacteria at the time of collection by RNA expression profiling
- Tube format is suitable for high-throughput analysis, increases efficiency and minimizes sample handling errors
- Compatible with downstream applications (e.g. RT-qPCR, microarray)
| Application | Target | Reference |
|---|---|---|
| Virus | DNA | EBV [1], HHV-8 [2,3], CMV [4] |
| RNA | HCV [5], HIV [5,6] | |
| Bacteria | DNA | HOMIM [7] |
| RNA | E. coli [9] | |
| Extraction systems | Abbott m2000 [5,6], Qiagen MinElute [8] | |
| Downstream applications | qPCR [1,2,3], Microarray [7] |
[2] High quality microbial DNA collected with OMNIgene•ORAL enables detection of HHV-8 in saliva. DNA Genotek. MK-00018.
[3] Evaluation of a new commercial kit for detection of human herpesvirus 8 (HHV-8) in saliva. http://www.dnagenotek.com/ROW/pdf/GriffithUniversity.pdf
[4] Genotyping of cytomegalovirus from toddlers’ saliva samples collected with OMNIgene•ORAL. DNA Genotek. MK-00050.
[5] Compatibility of saliva collected using OMNIgene•ORAL (OM-505) with the Abbott m2000rt RealTime System for the detection of viral RNA. DNA Genotek. PD-WP-00025.
[6] OMNIgene•ORAL (OM-505) stabilizes RNA from human viruses at room temperature for several weeks. DNA Genotek. PD-WP-00024.
[7] OMNIgene•ORAL stabilizes microbial DNA profiles in oral fluid samples, enables more precise characterization of oral flora. DNA Genotek. MK-00090.
[8] OM-505 Microbial DNA and RNA purification protocol using QIAGEN® QIAamp® MinElute Virus Spin Kit. DNA Genotek. PD-PR-00214.
[9] Internal validation using OM-505 extraction8 in house RT-qPCR.
More information .
-
Manufacturer websiteManufacturer website
-
Instruction manual (engl.)Instruction manual (engl.)
SARS-CoV-2 RNA detection from saliva .
To determine whether SARS-CoV-2 RNA can be reliably detected in saliva samples collected with OMNIgene-ORAL (OME-505) stored at room temperature, stability tests were performed on samples from 15 participants using an internally verified RUO assay optimized for the detection of SARS-CoV-2 RNA in saliva. Samples were spiked with heat-inactivated SARS-CoV-2 at two different concentrations depicting a low viral spike (500 cp/mL) and a high viral spike (2,500 cp/mL). Samples were stored at room temperature (23 ± 3°C) for 21 days and extracted at baseline and at the 7 and 21 day time points. The SARS-CoV-2 nucleocapsid gene (N1 and N2) was used to detect the stability of viral RNA by RT-qPCR. Samples with a cq value of <40 had detectable viral RNA. The human RNase P gene (RP) was included as a control for human nucleic acid detection.

Cq values for SARS-CoV-2 RT-qPCR targets N1, N2 (viral), and RNase P (human, RP) at day 0 (purple), day 7 (blue), and day 21 (orange) in low-positive (500 cp/ml, N = 15) and high-positive (2,500 cp/ml, N = 14) enriched saliva samples. The dotted line indicates the detection limit. If no amplification was detected, the cq was set to 45 (maximum cq) for visualization. Statistical significance was determined using a two-way ANOVA with Sidak test for multiple comparisons.
From: Colameco, S., Brown, A., & Crawford Parks, T. (2020). Ambient temperature stabilization of SARS-CoV-2 viral RNA in OMNIgene®-ORAL (OME-505) collected saliva samples. DNA Genotek https://www.dnagenotek.com/ROW/pdf/PD-WP-00069.pdf

ORE-100 .
Every expression analysis starts with the collection of RNA samples. ORAcollect® RNA (ORE-100) is an all-in-one system for the collection, stabilization and transport of RNA from saliva for in vitro diagnostics.

Advantages .
- Stabilizes samples during collection and ensures that DNA and RNA are unchanged at the time of processing
- Stabilizes human, viral and bacterial RNA from saliva at room temperature
- Reduces transportation and storage costs and complexity with up to 60 days stability of RNA at room temperature
- Easy self-collection of high-quality DNA and RNA from oral samples
- No refrigerated transports necessary
- Compatible with downstream applications (e.g. RT-qPCR, microarray)
- Format suitable for high throughput processing
More information .
-
Manufacturer websiteManufacturer website
-
Instruction manual (engl.)Instruction manual (engl.)

OM-501 .
The OMNIgene ORAL OM-501 kit enables the collection of high-quality samples for the molecular detection of microbial DNA. Samples are preserved well enough to avoid bias until analysis by microarrays or next generation sequencing.

Advantages .
- Improved patient compliance through painless, non-invasive specimen collection
- Increases specimen handling efficiency and reduces handling errors
- Ideal for on-site collection, transport at room temperature and long-term storage
- The sample remains stable for one year at room temperature, which reduces the cost and effort of transport and storage
- Format suitable for high throughput automated processing
| Venous blood | Mouthwash | Buccal swabs | OMNIgene•ORAL (OM-501) | |
|---|---|---|---|---|
| Non-invasive collextion | ✘ | ✘ | ✔ | ✔ |
| Standardized format for high-throughput processing | ✔ | ✘ | ✘ | ✔ |
| Specimen stability at room temperature | Days | Weeks | Days | 1 year |
| Blood collection | Oral collection |
More information .
-
Manufacturer websiteManufacturer website
-
Instruction manual (engl.)Instruction manual (engl.)
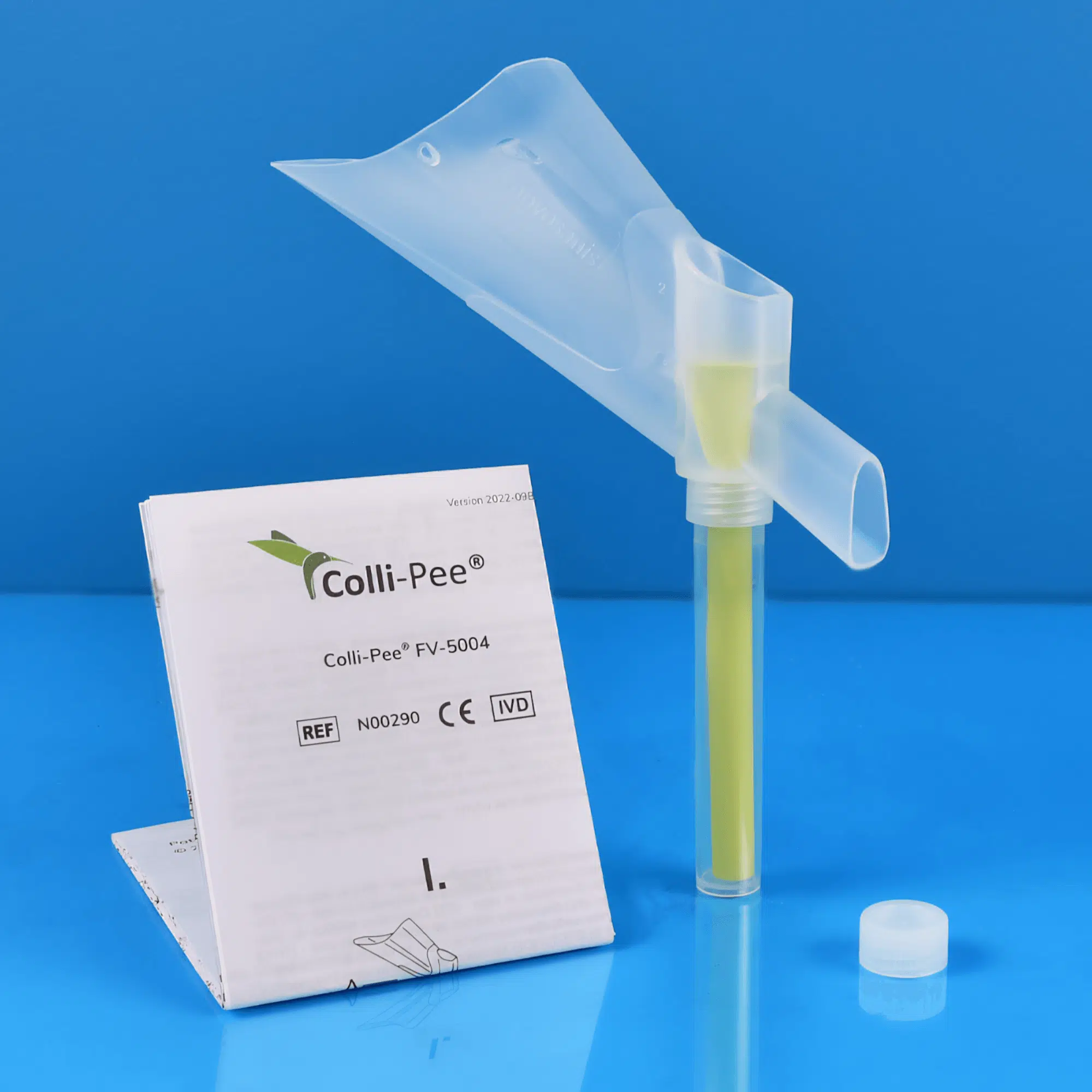
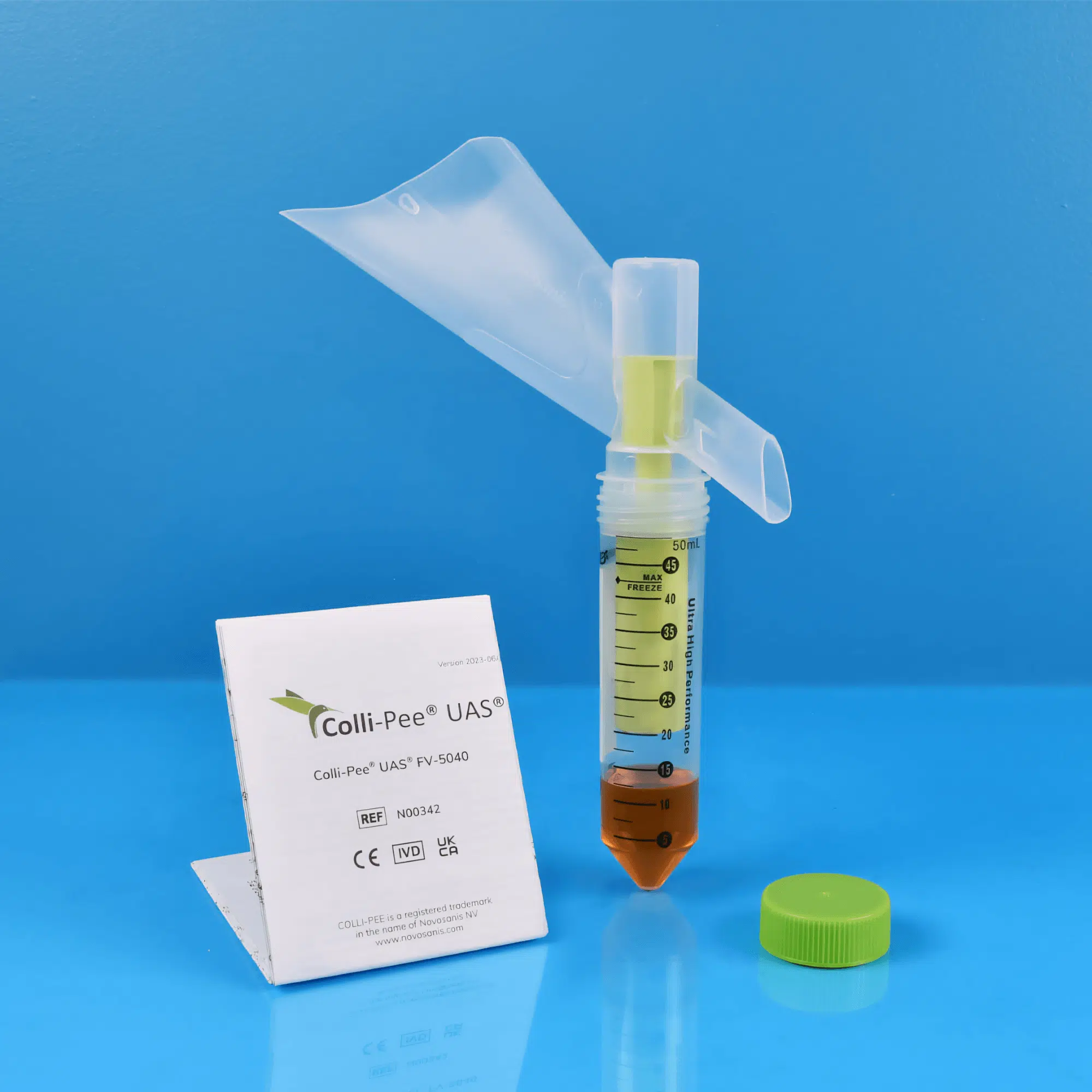
Colli-Pee® FV-5000 .
Colli-Pee® is a patented sampling device that allows volumetric and standardized collection of morning urine. The product range consists of variants that collect different volumes from 4 to 45 mL. The collection tubes can be pre-filled with a preservative, allowing for longer storage and/or shipment of urine at room temperature.
First urine contains higher concentrations of analytes associated with various sexually transmitted infections, including human papillomavirus (HPV) and Chlamydia trachomatis (CT) DNA. In addition, the collection of urine samples from the morning first stream is important for the identification of cancer biomarkers, e.g., for prostate cancer.

- Easy to use for both men and women
- Can be shipped to individuals as well as laboratories for testing purposes
- Sampling at home possible
- Optional: filling of Colli-Pee® with Novosanis' urine preservation medium (UCM) to preserve the DNA in the urine
-
Manufacturer websiteManufacturer website
-
Instruction manual (engl.)Instruction manual (engl.)
-
PublicationsPublications
Sampling for infectious diseases .
DNA and RNA sample collection from saliva or urine.